Question:
The sodium-independent iodide/chloride transporter is named as
- Megalin
- Pendrin
- Transthyretin
- Prestin
Rephrasing the question:
What is the name of the sodium-independent iodide/chloride transporter?
Answer:
Pendrin is the sodium-independent iodide/chloride transporter (option 2)
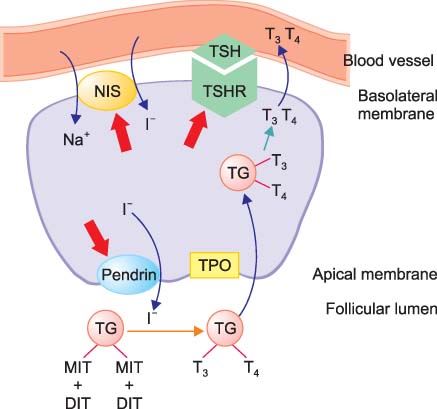
Explanation:
Let’s go through and discuss each of these proteins:
- Megalin
Megalin is also known as LRP2 (low density lipoprotein-related protein 2). It is a receptor found in the plasma membrane of many absorptive epithelial cells that binds to multiple ligands and mediates their endocytosis. Megalin is involved in proximal tubule protein uptake in the kidney, and serves as a receptor for the protein thyroglobulin in thyrocytes (epithelial cells in the thyroid). - Pendrin
Pendrin is an anion exchange protein, interesting as a sodium-independent chloride-iodide exchanger. It controls the flow of iodide into the thyroid gland. Mutations in this protein are associated with Pendred syndrome, the most common form of syndromic deafness, an autosomal-recessive disease. Pendred syndrome is characterized by thyroid goitre and enlargement of the vestibular aqueduct resulting in deafness. It also seems to have a role in airway hyperreactivity and inflammation, possibly due to pendrin’s effects on ion concentration in the fluid in the airways. - Transthyretin
Transthyretin is a transport protein found in the serum and cerebrospinal fluid that transports thyroxine and retinol. The transthyretin in the blood is formed in the liver, and that in the cerebrospinal fluid is secreted by the choroid plexus. Transthyretin misfolding and aggregation is associated with amyloid diseases such as senile systemic amyloidosis, familial amyloid polyneuropathy, and familial amyloid cardiomyopathy. Considering this, it is not surprising that functional transthyretin also has a protective effect in Alzheimer’s disease. - Prestin
Prestin is the motor protein of the outer hair cells of the inner ear of the mammalian cochlea. It is critical to sensitive hearing in mammals, and mutations in it are associated with non-syndromic hearing loss. Recently, anion transport has been shown to be an important mediator of prestin’s effect on the electromotility of hair cells.
Sources:
https://en.wikipedia.org/wiki/LRP2
https://en.wikipedia.org/wiki/Pendrin
Fong, P. Thyroid iodide efflux: a team effort? The Journal of Physiology, 589(24): 5929-5939.
https://www.creative-biolabs.com/slc26a4-membrane-protein-introduction.html

Very Well Made !!👏👏
I really appreciate that!😄😃
i have learn several just right stuff here. Definitely value bookmarking for revisiting.
I wonder how so much effort you place to make one of these fantastic informative web site.
온라인카지노” m really impressed with your writing skills and
We finalize our work space and hamper within your budget
, no matter what kind of programme you have in mind!
hi / hello there
i have learn several just right stuff here. “밤의부산”Definitely value bookmarking for revisiting.
I wonder how so much effort you place to make one of these fantastic informative web site.
thank you i love it.
You have somke reaⅼly good posts and I belіeve I would bе a gooⅾ asset. If yoᥙ ever wɑnt too take somе oof
tһe load off, Ι’d absolutеly love카지노사이트 tⲟ ѡrite sߋme c᧐ntent for your
blog in exchange for a link Ьack to mіne. Please send me an email іf іnterested. Tһank ʏou!
Appreciation for taking the time to discuss this topic, I would love to discover
more on this topic. If viable, as you gain expertise, would you object to updating
the website with further information?“강남안마” It is tremendously beneficial for me.
Fantastic goods from you, man. I have understand
“온라인바카라”
your stuff previous to and you’re just too fantastic.
on a daily basis. It will always be interesting to read through content from
other authors and practice a little something from other sites.
온라인바카라
Slotv9ผู้ ให้บริการ เกมสล็อตออนไลน์ ลำดับที่หนึ่ง เกมสล็อตออนไลน์ มาแรง 2022 สล็อตออนไลน์ pg น่าเล่น เกมใหม่ มาแรง ที่พวกเรา ได้คัดสรร มารวมไว้ ที่เดียวให้ท่าน ได้สัมผัส
Marine88เป็นเว็บไซต์ที่เชี่ยวชาญด้านการเดิมพันกีฬาและ PG SLOTออนไลน์ ซึ่งมีการบริการที่มีคุณภาพและปลอดภัยสำหรับผู้เล่นทุกระดับ ด้วยระบบการเงินที่มั่นคงและรวดเร็ว
land slot auto เล่นเกมสล็อตได้ทุกค่าย ครบทุกเกมไปกับ ทุกค่ายเกมสุดสนุก แบบแปลกใหม่ ไม่มีซ้ำใครกันแน่ เล่นได้ไม่น่าเบื่อ PG ไม่เหมือนกันด้วยการเป็นบริการเกมสล็อตออนไลน์
zeed456 walletเป็นสินค้าที่มีคุณภาพดีและน่าสนใจมากๆ โดยเฉพาะอย่างยิ่งสำหรับคนที่ต้องการความสะดวกสบายในการใช้งาน PG SLOTและการเดินทาง ด้วยการออกแบบที่ดี
pg slot 168galaxy ยอดเยี่ยมที่เกมที่มีระบบระเบียบทำการระดับประเทศแบบอย่างเกมน่าเล่นเกมแนวหน้าที่สมัยบริการ24 ช.ค่ายเกม pg slot มาแรงและเป็นค่ายเกมที่มีผู้เล่นเข้าเล่นเยอะ
Riches888 Allถือเป็นหนึ่งในเกมส์คาสิโนออนไลน์ที่มีชื่อเสียงและความนิยมสูงในวงการเดิมพันใน PG SLOTออนไลน์ โดยได้รับการพัฒนาและออกแบบโดยทีมงานมือ
ทาง เข้า สล็อต pg เกมสล็อตออนไลน์ พร้อมที่ จะพาทุกท่าน PG SLOT บันเทิงใจ ในลักษณะของ เกมส์ บนโทรศัพท์ มือถือ และก็ยังไม่หมด เพียงเท่านี้ ในเกมนั้นยังมี โบนัส ก้อนโตรออยู่
เกมApollo PG Slotเป็นเกมส์สล็อตออนไลน์ที่ถูกพัฒนาโดยบริษัท PG SOFT™ ที่มีความเชี่ยวชาญทั้งในด้านภาพและเทคโนโลยีสูงสุด เกมส์สล็อตของ PG SLOT
slotการตรวจสอบอัตราการจ่ายและรางวัลก่อนเล่น เพราะ การเดิมพันสูงสุดไม่ใช่ตัวเลือกที่ถูกต้องเสมอไป เล่นเกม Pg Slot เฉพาะเกมส์ที่ตัวเองรู้จักดี
การใช้ crow789เมื่อไหร่จะถือว่าชื่อนี้เป็นชื่อที่เหมาะสมหรือไม่นั้นขึ้นอยู่กับวัตถุประสงค์และหลักการของคุณใน PG SLOTเองว่าต้องการเลือกใช้ชื่อในการติดต่อสื่อสารแบบนี้หรือไม่
ทำเงินกับเกมสล็อตออนไลน์ให้ได้ง่ายมากยิ่งขึ้น ด้วยเเผนเด็ด กลยุทธ์ เล่นสล็อต ซึ่งไม่ว่าใครก็สามารถทำเงินกับเกมสล็อตออนไลน์ได้ง่ายยิ่งขึ้น มือใหม่เล่นได้สบายๆ เพราะว่าถ้าผู้เล่นมีเเผนที่ดีก็มีชัยไปกว่าขึ้น ทำเงิน สร้างกำไรกับเกมสล็อตออนไลน์ด้วยกลยุทธ์ฟรี ที่เราได้จัดทำขึ้น เเจกให้ทุกคน เเจกแบบฟรีๆ
recommend website to play online slots make real money Unlimited withdrawals Transactions. nagagame
That’s really nice. the new website to play online slots. ทดลองเล่นสล็อต
I like that.pg slot game with the complete strategy listed on them that you can use. ทดลองเล่นสล็อตซื้อฟีเจอร์
It affects men’s voluptuous power gravely and leaves them온라인바카라 unsatisfied during those hidden moments. Infirm carnal life-force of men is a serious problem. a new standard for unforgettable gaming
เดิมพัน ค่ายเกม พีจีสล็อต โบนัส แตกบ่อยเนื่องจากจุดเด่นสำหรับการเล่นเกมต่างถูกกล่าวขานรวมทั้งบอกต่อกันมากมายก่ายกอง pg slot จากผู้เล่นซึ่งสามารถปราบรางวัลข้างในเกมมากมาย
PG SLOT 11ป็นเว็บไซต์เกมสล็อตออนไลน์ที่ได้รับความนิยมอย่างมากในปัจจุบัน มีตัวเลือกให้เล่นมากมายและตอบโจทย์การเล่นเกม PGSLOT
pg สล็อตแตกง่าย ค่ายเกมสล็อตออนไลน์ ที่ได้เป็นขวัญใจของผู้เล่น รวมทั้งนักปั่นสล็อตตัวจริงมากที่สุดในตอนนี้ สล็อตค่าย pg แตกง่าย เกมสล็อตรูปแบบใหม่ ตอบปัญหาผู้เล่นมากยิ่งกว่าเดิม
PG Slot Game888?เป็นเกมสล็อตออนไลน์ที่มีความน่าสนใจสำหรับผู้เล่นที่ชื่นชอบการเสี่ยงโชค โดยเกมนี้มีการออกแบบให้มีความสวยงามและมีความเข้ากันได้ใน PGSLOT
mega slot
megagame slot
สล็อตเดโม่ฟรี
naga game slot
Every game, Every gamer, All in one place. Welcome to Lucky Cola
Join the ultimate gaming community on and conquer virtual worlds together! Hawkplay
Epic Quests, Your Way: Conquer foes and create your gaming saga. Lucky Cola
imba96ออนไลน์ ยังมีการบริการลูกค้าอย่างมืออาชีพ ที่พร้อมให้คำปรึกษาและแก้ไขปัญหาต่างๆ pg slot ให้กับผู้เล่นโดยตลอด ไม่ว่าจะเป็นการสอบถามด้านเทคนิค การเลือกเล่นเกมส์
สล็อต pg ใหม่ ล่าสุด 2024 อัพเดทเกมไวก่อนใครทดลองเล่นสล็อต สล็อตpg 2023 ใหม่ รวดเร็วทันใจ เรียกได้ว่าอัพเดทกันแบบเรียงไทม์ ค่ายเกม ลงเกมหมดปุ๊บ ก็สามารถเข้ามา ทดลองเล่นสล็อต pg ใหม่ กับเราได้ทันที เพราะเราเป็นพาร์ทเนอร์ กับหลายๆค่าย
เว็บ 2024 เล่นฟรีเว็บใหญ่ทำให้สามารถนำเกมใหม่มาลงให้คุณ pgslot-game.app/ ได้เล่นก่อนใคร เพียงแค่คุณคลิกเข้าเว็บเราในหน้า ทดลองเล่นสล็อต ก็สามารถที่จะคลิก เพื่อเล่นเกมได้เลยก่อนใคร ไม่จำเป็นจะต้องสมัครสมาชิก ให้ยุ่งยาก ก็สามารถเข้ามาเล่นสล็อต ได้แบบฟรีๆ พร้อมเครดิตฟรี ให้คุณได้หมุนกันแบบจุใจ และมีแจกใหม่เรื่อยๆ
สล็อตออนไลน์ แหล่งรวมเกมเดิมพันบนมือถือ ทุกเกมได้แบบไม่มีสะดุด landslot.info พร้อมทั้งยังสามารถ ที่จะกดเข้ามาเพื่อสมัครสมาชิกกันแบบฟรี หากทดลองเล่นสล็อต แล้วอยากเล่นจริงก็สามารถ สมัครได้เลยแบบง่ายๆ ใช้เวลาไม่เกิน 1 นาทีก็สามารถเติมเงิน ด้วยระบบออโต้
เปิดเว็บใหม่ล่าสุด เข้ามาเล่นกับเราได้เลย ทดลองเล่นสล็อต pg ทั้งหมด สล็อตเว็บตรง โดยตัวระบบออโต้นั้น ใช้เวลาในการแอดเงินเข้าระบบ ให้ท่านหลังจากโอนเงินโดยใช้เวลาเพียงไม่เกิน 1 นาที
แจกเครดิตฟรีเล่นเกมออนไลน์ของเรา เรียกได้ว่ารวดเร็วทันใจ ไม่มีอารมณ์เสียอย่างแน่นอน ทดลองเล่นสล็อต pg ไม่ เด้ง ซื้อฟรีได้ สล็อตทดลองเล่น เล่นฟรี พร้อมอัพเดทเกมใหม่ล่าสุดรับปี 2024
เกม slotxo เป็นเกมสล็อตสล็อตรูปแบบแรก ที่ได้พัฒนามาจากการเล่นสล็อตผ่าน ตู้สล็อตแมชชีน ผู้เล่นจึงสามารถทำความเข้าใจรูปแบบการเล่นได้ง่าย แม้จะเป็นผู้เล่นมือใหม่ ก็สามารถทำความเข้าใจในเรื่องของกฎ กติกาได้อย่างรวดเร็ว นอกจากนี้ เกมสล็อตจากค่ายเกมนี้ ยังมีเนื้อเรื่องเกี่ยวกับ วัฒนธรรม เข้ามาสอดแทรก เพื่อให้ผู้เล่นได้รับ อรรถรสความสนุก ในการเล่นเกมมากยิ่งขึ้น สิ่งที่ทำให้ขาดทุนจากการเล่นเกมslot
สล็อต ufa789 เว็บพนันครบวงจร แทงบอล บาคาร่า สล็อต ครบจบที่เดียว
THE ONE CARGO ชิปปิ้ง นำเข้าสินค้าจากจีน ครบวงจร ที่ได้มาตรฐาน และมีระบบทันสมัยรองรับความต้องการของลูกค้าทุกรูปแบบ ขอให้เราได้ช่วย เรารับประกันว่า เราจะทำให้ดีที่สุดเพราะความพึงพอใจของลูกค้าคือกำไรของเราเรามีทีมงานที่เชียวชาญทุกด้าน
I admire your creativity in problem-solving. You bring fresh ideas to the table that inspire us all. Check my page for other related articles ipl 365
Hey I know this is off topic but I was wondering if
you knew of any widgets I could add to my blog that automatically tweet my newest twitter updates.
I’ve been looking for a plug-in like this for quite some time and was hoping maybe you would have some exp구미출장샵erience
Uncover the secrets of the ultimate online game Hawkplay
THE ONE CARGO บริการ drop shipping นำเข้าสินค้าจีน มาไทย เรามีโกดังเป็นของตัวเอง และมีพนักงานคอยรับสินค้า เพื่อคัดแยกสินค้าจากทางรถและทางเรือ โดยส่งสินค้าทุกวัน สำหรับลูกค้าที่สั่งสินค้าเองและต้องการคนรวบรวมของ เรามีบริการแบบครบวงจร
I admire your creativity in problem-solving. You bring fresh ideas to the table that inspire us all. Check my page for other related articles ipl tata
I admire your creativity in problem-solving. You bring fresh ideas to the table that inspire us all. Check my page for other related articles ipltata
This helped a lot, thanks!
Very informative, thanks!
You’ve done a great job here—your hard work really shows! kinggame digital
Great job! This is a clear and concise explanation. kingph casino
การมี ระบบขายของออนไลน์ เข้ามาเป็นระบบหลังบ้านให้กับร้านค้า จะช่วยทำให้งานต่างๆ เป็นระเบียบมากยิ่งขึ้น เพราะการขายของออนไลน์ เป็นเรื่องที่ยุ่งยากมากหากทำด้วยตัวคนเดียว ต้องโฟกัสกับการขาย เช็คสต๊อก แพ็ค และส่ง ถ้ามีการทำทั้งหมดนี้ด้วยตัวคนเดียว ก็ถือเป็นเรื่องที่เหนื่อยสุดๆ แต่หากมีการนำโปรแกรมขายของออนไลน์เข้ามาช่วย ก็จะทำให้เรื่องยุ่งยากพวกนี้เป็นเรื่องที่ง่ายขึ้น
Your videos always bring such positive energy! I really appreciate the effort you put into each one, and it’s clear that you love what you do. Keep killing it! 🙌❤️
piso game
This is really interesting, You’re a very skilled blogger. I have joined your rss feed and look forward to seeking more of your great post. Also, I have shared your website in my social networks! lsm99 ฝาก ถอน
Easy investment, get real money at tga69, a stable platform ready to take care of you 24 hours a day. https://tgabet69.co/
Interested in starting without risk? Come here. Try playing slots for free without deposit. Have fun immediately. No need to use capital first. https://ambslot.land/
Enjoy quality games from pg slot club. Easy to play, bonus breaks quickly. Apply today and receive special privileges immediately. Don’t miss it. https://pgclub.io/
Come try out the free trial slots before anyone else. No expenses. Have fun every day here. Get a chance to win many prizes. https://naga.live/
Enjoy quality games from pg ink. Easy to play, lots of bonuses, with special promotions. Apply today and receive immediately. https://inkspin.org/
Sign up today at https://nagagames-th.com/ Enjoy a variety of games with many special promotions waiting for you here.
Try playing naga game today, super fun with bonuses and big prizes. Easy to register, get free credit immediately. https://nagagames-th.io/
Easy to apply and enjoy exciting games at https://www.nagagames-th.org/ along with receiving special bonuses and many benefits.
Sign up now at tgabet to experience the ultimate betting experience with many bonuses and promotions waiting for you. https://tgabet-22.info/
Have fun playing games at tga slots, enjoy big bonuses and many prizes, with special promotions waiting for you. https://tgabet89.net/